Postdoctoral Associate
phone: 531-346-4648
EDUCATION
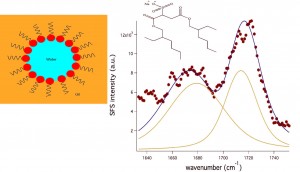
Emulsions are made up of either water droplets dissolved in oil or oil droplets dissolved in water stabilized by ampiphillic surfactant molecules. They are important for the food and cosmetic industry, water remediation, and nanoparticle synthesis. We would like to understand their local chemical environment at the oil water interface in order to gain insight in their formation and stabilization. Sum Frequency Scattering (SFS) allows the vibrational modes of molecules at the curved oil water interface to be probed where further information about their orientation and assembly can be determined on a molecular level.
I am interested in understanding the behavior of sodium dioctyl sulfosuccinate (AOT) at the water droplet/ CCl4 interface by probing the carbonyl head group using vibrational sum-frequency spectroscopy in both the planar and scattering geometry. The emulsions studied so far have been around ~200 nm in diameter. By comparing the sum-frequency of the curved and planar oil/water interfaces, we can find interesting information about the differences in head group solvation environment, chain disorder and water confinement.