We are interested in the chemistry of soils, sediments, aquifers, and other environments under both natural and contaminated settings. The chemistry in these environments is complicated, due to the overlapping controls by diverse physical, chemical, and biological processes. Our research questions range from theoretical to practical, from the fate of arsenic and other contaminants to the influence of environmental chemistry on biological processes. Our goal is to elucidate and predict how environmental chemistry evolves with time across different spatial and temporal scales.
Our approach
We combine laboratory and field experiments with numerical modeling to address questions of environmental chemistry. Our experiments test natural environments directly in the field or indirectly in laboratory reactors. As the experiments progress, we collect a suite of chemical parameters, including those of gasses, aqueous solutions, and solid minerals. Our routine analyses include gas chromatography (GC), ion chromatography (IC), high-performance liquid chromatography (HPLC), and spectrophotometer. Our goal is to monitor closely and comprehensively how the chemistry evolves with time.

Running a push-pull test in an arsenic-contaminated aquifer
Our modeling efforts cover a wide range of scales, from pore fluids to field-scale reactive transport. The goal is to summarize the current understanding of geochemical and biogeochemical processes in the environment, to explore numerically the gap between our current understanding and the complexity of natural systems, and to identify critical processes that control reaction paths of geochemical and biogeochemical systems.
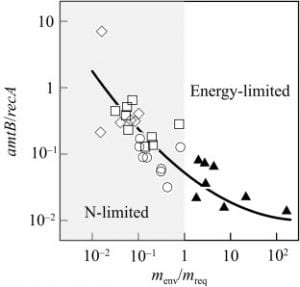
Nitrogen- vs energy-limited microbial growth during an in situ bioremediation aquifer test